
It’s often helpful to keep track of our atom count in a table. If we count the atoms on each side of the reaction, we’ll discover that they are not equal! While this equation accurately represents the chemical identity of the reactants and products, it’s unbalanced. Methane reacts with oxygen to produce carbon dioxide and water. Balancing Equations Examplesįor example, here’s the unbalanced reaction of the combustion of methane. Conversely, an unbalanced reaction has unequal numbers of each type of atom on the reactant and product side of the equation. Chemists represent this in a balanced chemical equation, in which the number of each type of atom on the reactant side equals the number on the product side. In other words, when reactants recombine or decompose to form products, we can’t lose or create atoms- we have to end with the same number we started with. In any reaction, the law of conservation of mass must be followed. 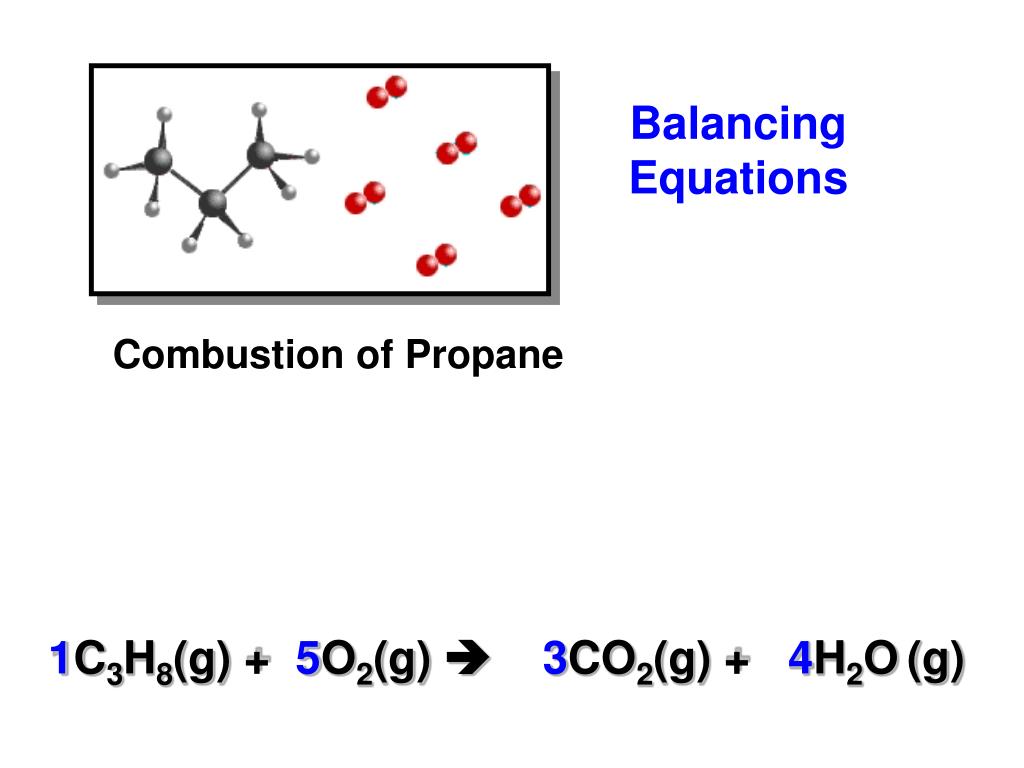
Hi, and welcome to this video on balancing chemical equations!
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
